Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -dialkylamides using multistage mixer-settler extractors
-dialkylamides using multistage mixer-settler extractorsBan, Yasutoshi; Hotoku, Shinobu; Tsutsui, Nao; Suzuki, Asuka; Tsubata, Yasuhiro; Matsumura, Tatsuro
Procedia Chemistry, 21, p.156 - 161, 2016/12
Times Cited Count:5 Percentile:94.32(Chemistry, Inorganic & Nuclear)A continuous counter-current experiment was carried out to demonstrate the validity of a process using  -dialkylamides for recovering U and Pu. This process consisted of two cycles, and the 1st cycle and the 2nd cycle employed
-dialkylamides for recovering U and Pu. This process consisted of two cycles, and the 1st cycle and the 2nd cycle employed  -di(2-ethylhexyl)-2,2-dimethylpropanamide and
-di(2-ethylhexyl)-2,2-dimethylpropanamide and  -di(2-ethylhexyl)butanamide as extractants, respectively. The feed solution for the 1st cycle was 5.1 mol/dm
-di(2-ethylhexyl)butanamide as extractants, respectively. The feed solution for the 1st cycle was 5.1 mol/dm (M) nitric acid containing 0.92 M U, 1.6 mM Pu, and 0.6 mM Np. The raffinate collected in the 1st cycle was used as the feed for the 2nd cycle. The ratios of U recovered in the U fraction and U-Pu fraction were 99.1% and 0.8%, respectively. The ratio of Pu recovered in the U-Pu fraction was 99.7%. The concentration ratio of U with respect to Pu in the U-Pu fraction was 9, and this indicated that Pu was not isolated. The decontamination factor of U with respect to Pu in the U fraction was obtained as 4.5
(M) nitric acid containing 0.92 M U, 1.6 mM Pu, and 0.6 mM Np. The raffinate collected in the 1st cycle was used as the feed for the 2nd cycle. The ratios of U recovered in the U fraction and U-Pu fraction were 99.1% and 0.8%, respectively. The ratio of Pu recovered in the U-Pu fraction was 99.7%. The concentration ratio of U with respect to Pu in the U-Pu fraction was 9, and this indicated that Pu was not isolated. The decontamination factor of U with respect to Pu in the U fraction was obtained as 4.5 10
10 . These results supported the validity of the proposed process.
. These results supported the validity of the proposed process.
Ban, Yasutoshi; Hotoku, Shinobu; Tsutsui, Nao; Suzuki, Asuka; Matsumura, Tatsuro
Proceedings of 5th International Conference on Asian Nuclear Prospects 2016 (ANUP 2016) (CD-ROM), 2 Pages, 2016/10
A continuous counter-current experiment was conducted using mixer-settler extractors installed in a hot cell.  -Di(2-ethylhexyl)-2,2-dimethylpropanamide (DEHDMPA) was used as the extractant for U. The experimental results showed that DEHDMPA can effectively extract U from nitric acid, and the recovery of U in the U fraction was obtained as 99.6%.
-Di(2-ethylhexyl)-2,2-dimethylpropanamide (DEHDMPA) was used as the extractant for U. The experimental results showed that DEHDMPA can effectively extract U from nitric acid, and the recovery of U in the U fraction was obtained as 99.6%.
 and UO
and UO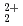 ions with tridentate ligand diglycolamide (DGA)
ions with tridentate ligand diglycolamide (DGA)Hirata, Masaru; Guilbard, P.*; Dobler, M.; Tachimori, Shoichi
Physical Chemistry Chemical Physics, 5(4), p.691 - 695, 2003/02
Times Cited Count:20 Percentile:54.61(Chemistry, Physical)no abstracts in English
Morita, Yasuji; Sasaki, Yuji; Tachimori, Shoichi
Proceedings of International Conference on Back-End of the Fuel Cycle: From Research to Solutions (GLOBAL 2001) (CD-ROM), 7 Pages, 2001/09
no abstracts in English
Morita, Yasuji; Yamaguchi, Isoo; ; *; Kubota, Masumitsu
Proc. of the Int. Conf. on Future Nuclear Systems (GLOBAL'99)(CD-ROM), 8 Pages, 1999/00
no abstracts in English
Hirata, Masaru; Sekine, Rika*; Onoe, Jun*; Nakamatsu, Hirohide*; Mukoyama, Takeshi*; *; Tachimori, Shoichi
Journal of Alloys and Compounds, 271-273, p.128 - 132, 1998/00
Times Cited Count:8 Percentile:52.63(Chemistry, Physical)no abstracts in English
; Tachimori, Shoichi
Bulletin of the Chemical Society of Japan, 70(1), p.135 - 142, 1997/00
Times Cited Count:2 Percentile:19.46(Chemistry, Multidisciplinary)no abstracts in English
; ;
Journal of Nuclear Science and Technology, 16(6), p.434 - 440, 1979/00
Times Cited Count:16no abstracts in English
Mikrochim.Acta,1976 II, (5-6), p.559 - 568, 1976/00
no abstracts in English
; ;
Bunseki Kagaku, 18(2), p.208 - 213, 1969/00
no abstracts in English
Dai-8-Kai Nihon Aisotopu Kaigi Hobunshu, B-(2)-4, p.260 - 262, 1965/00
no abstracts in English
; Umezawa, Hirokazu;
Nihon Genshiryoku Gakkai-Shi, 1(5), p.299 - 303, 1959/00
no abstracts in English
Sasaki, Yuji; Matsumiya, Masahiko*
no journal, ,
In our laboratory, the mutual separation of lanthanides by batch-wise multi-stage extraction is conducted. Dy is introduced into Nd-magnet, for this recycle mutual separation of Dy and Nd in important. Here, the solvent extraction method is used for their separation. We confirm the consistency between calculation and experimental results and perform the separation experiments of Dy from Nd. The results will be shown in the presentation.
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
no journal, ,
In our group, the separation of An from Ln is performed using batch-wise multi stage extraction. The condition, DGA(diglycolamide)-DTPA(diethylenetriamine-pentaacetic acid) or DGA-DTBA(diethylenetriamine-triacetic diamide),showed approximate 10 of D(Am)/D(Ln). The multi stage extraction of 10  10 of organic and aqueous phase numbers gives successfully 97% of Am with 3% of Ln in the aqueous phase.
10 of organic and aqueous phase numbers gives successfully 97% of Am with 3% of Ln in the aqueous phase.
Nomizu, Daiki; Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Katsuta, Shoichi*
no journal, ,
In our group, the mutual separation of lanthanides by solvent extraction method is conducted. We focus on two phases in solvent extraction, the hydrophilic and lipophilic reagents are employed in both phases. Namely, using the condition of TODGA in organic phase and DOODA(C2, dioxaoctane-diamide) in aqueous phase or DOODA(C8) in organic phase and TEDGA in aqueous phase, the distribution ratios of Ln are compared. Using these results, the batch-wise multi stage extractions are performed in order to separate light and heavy Ln.
長縄 弘親; 永野 哲志
not registered
【課題】正抽出部100、洗浄部200、及び逆抽出部300が一体となり同期して機能する循環送液システムにおいて、複数回の循環回数によって発現する多段に相当する効果を利用して分離精製される特定の物質の製造方法を提供すること。 【解決手段】正抽出部100、洗浄部200、及び逆抽出部300が一体化して同期的に機能するシステムを用いて、正抽出部100の水相(多くの場合、重液相)が正抽出部100のみの単独部内で循環送液すると同時に、正抽出部100の油相(多くの場合、軽液相)が洗浄部200から逆抽出部300を経て再び正抽出部100に至る横断的な循環送液を行うように制御し、正抽出部100の水相の単独部内循環を複数回行うことによって分離精製される特定の物質の製造方法。